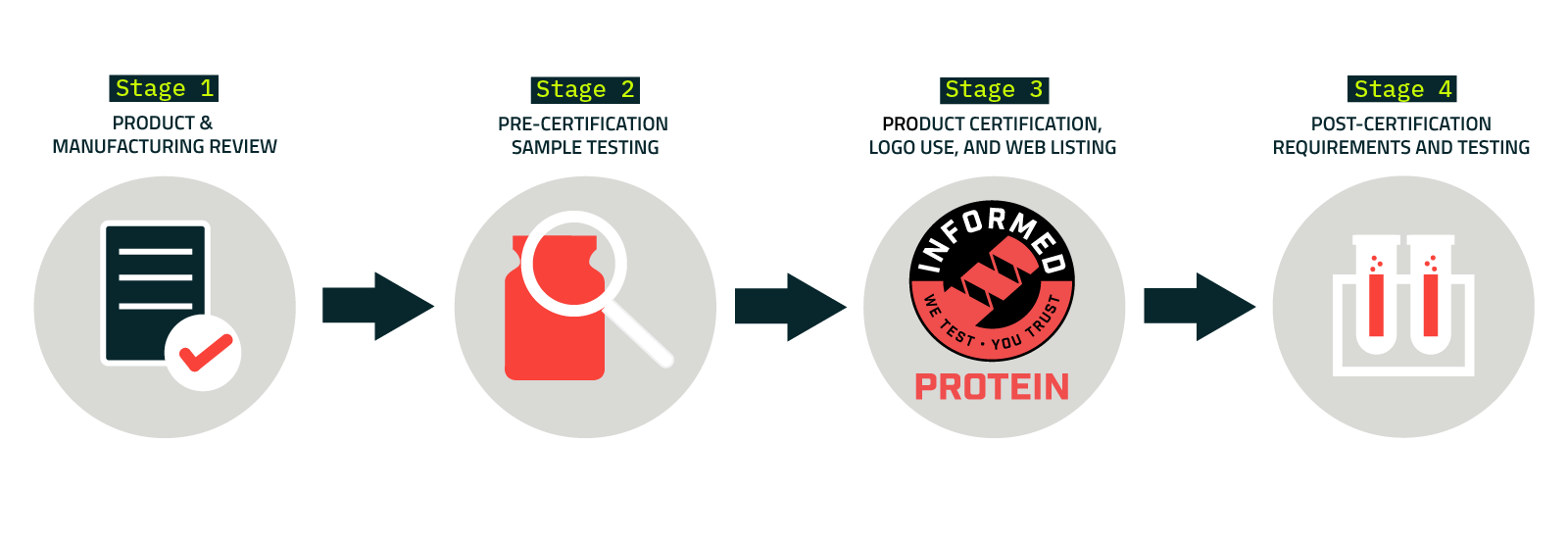
Sports nutrition and dietary supplement products certified by Informed Protein undergo a rigorous lab-tested protein supplement certification process, consisting of four stages:
Ready for your products to join our globally recognized protein verification programme? Click below to contact us and begin the path to Informed Protein certification.
Stage One: Product & Manufacturing Review
Prior to protein certification, a certification agreement will be issued along with a programme pack explaining the programme. Organisations can also send product labels to the Informed Protein marketing team for approval though no products bearing the Informed Protein logo can be sold until the entire protein certification process is complete.
A comprehensive picture of the product is built up and reviewed to understand factors such as product variants, retail plans, product testing schedules, and manufacturing facilities. At this stage, the product formulations are reviewed in detail by experienced assessors to establish the types of protein used, the amount of protein included in the formulation, and protein levels on the packaging label.
A review of the procedures in place at relevant manufacturing facilities is then undertaken via a paper-based assessment. This process is managed by experienced assessors, who provide guidance on how to meet the required standards of the Informed Protein programme.
This stage of the protein certification process includes a manufacturer assessment of the following:
- Quality systems and manufacturing GMP or equivalent audits
- Staff training in relation to cross-contamination prevention
- Raw material supplier assessment procedures
- Traceability & recall procedures
Stage Two: Pre-Certification Sample Testing
Prior to Informed Protein supplement certification, we test three samples across a minimum of three production runs/batches. All samples are tested at our protein testing labs for total protein (via the determination of total nitrogen), total carbon, free amino acids, and a range of nitrogen-rich adulterants (e.g. melamine and associated analogues). All results must meet label claim requirements for protein.
Heavy metal and pesticide testing must also be carried out by a qualified laboratory and submitted for review.
This stage will be carried out in parallel with the product and manufacturing review (stage one).
Stage Three: Product Certification, Logo Use, and Product Page
Once stages one and two are complete and any resulting actions have been closed out, the product is accepted for Informed Protein certification. An Informed Protein certificate will be issued and the company can use the Informed Protein logo. Companies earning certification also receive the 'Informed Protein Brand Standards' which explain how to use the logo to promote your certification and maximise return on investment. Each certified product also has its own product page at wetestyoutrust.com to support marketing and allow consumers to verify the certification status of products with the mark on the label.
Stage Four: Post-Certification Requirements and Testing
Following Informed Protein certification, each product must undergo a minimum of monthly blind testing to independently ensure the integrity of the certified product. Blind samples are purchased by the Informed Protein Programme Management team from online and physical retail stores and sent to the laboratory for testing.
The tested batches will be listed on the Informed Protein website on a page dedicated to the certified protein brands. In addition to routine protein supplement testing, production processes and manufacturing facilities are regularly re-assessed to ensure ongoing programme compliance.
If you’ve ever wondered what are the benefits of third-party testing protein, our overview page explains how certification ensures label accuracy, product safety, and consumer trust.






